| |
|
Inside this issue:
- Biofilm.
- Informed Consent.
We trust you are all well and have been enjoying the lovely weather we have been having around the country.
This is the first newsletter of 2019 and I hope it will be of interest to you.
Remember - if you have a topic you want to be covered or any have questions email me, Jane on [email protected]
|
|
BIOFILMA biofilm is a structured community of self-protecting, nonplanktonic, and often dormant bacteria, that tightly adhere to both animate and inanimate surfaces via a self-producing extracellular polymeric matrix (EPM); a substance that internally adheres whilst being externally "slimy".
Bacteria of all kinds gather and deposit on surfaces, secreting the polysaccharide (bound sugars) substance that becomes the encompassing biofilm matrix.
The bacteria within the biofilm thrive on degraded tissue in the case of wounds, by-products of its own extracellular polymer matrix, and in the case of endoscopes, on newly introduced bacterial substrate.
"A biofilm is a structured community of self-protecting bacteria that tightly adhere to both animate and inanimate surface"
Bacteria within the biofilm send out signals to call in more bacteria, which respond and are converted from mobile planktonic metabolically active bacteria to exuding the same phenotype (observable characteristics) as the bacteria within the biofilm: quiescence and immobility; they join the growing biofilm community.
This signalling process is called quorum sensing.
Additionally, the then mature biofilm will shed some of its mass which relocates to other areas on the same, or different surface, and the process of subsistence continues.
"Once mature a biofilm will shed some of its mass which relocates to other areas on the same, or different surface, and the process of subsistence continues"
Due to the matrix covering the bacteria, and to the quiescent (dormant) state of the non-planktonic bacteria, antibiotics and antimicrobials do not often work to remove or break down the biofilm.
Physical breakdown of biofilm, by way of manual cleaning using surfactant containing solutions, is essentially the only successful way of removing it, however, any residue can replenish within 24-72 hours; hence the rationale behind 24 hour stand down micro testing protocols.
Within this 24-72 hour window the disrupted biofilm leaves bacteria free to convert back to its planktonic form, and therefore unprotected by an extracellular polymer matrix and susceptible to antibiotics and antimicrobials.
"Any residue left behind can replenish within 24-72 hours"
Biofilms will eventually cause an increase in bioburden, and in the case of endoscopy can then redistribute itself onto the surfaces it comes into contact with - the intestine.
Increased bioburden creates localised infection, which in today's era of antimicrobial resistance (AMR), is extremely concerning; especially when we consider that two of the carbapenem-resistant Enterobacteriaceae (CRE) - Klebsiella and Escherichia coli - are found within the gut and have been implicated in endoscopic related infection outbreaks in recent years.
|
|
|
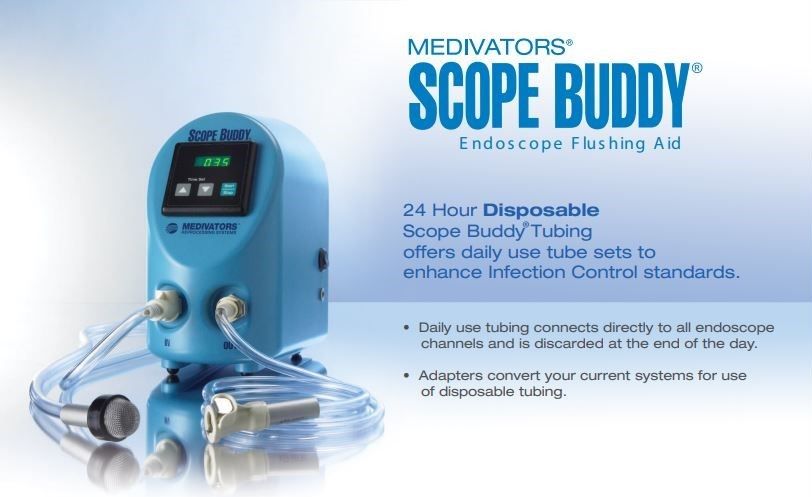
The continued moistening and drying of an endoscope - necessary for both use and reprocessing - can enhance biofilm growth; for this reason, pre-cleaning (the bedside clean especially) needs to be performed promptly and well to remove as much bioburden and bacterial "soil" as possible before the endoscope enters disinfection: disinfection alone will not remove biofilm.
Recent advances in channel cleaning technology as seen with the Pull-Thrusee a distinct improvement in the removal of bioburden over traditional brush techniques. The combination of the physical disruption caused by the disk-based clean, and the suction pull through of detergent, looks promising in regards to decreasing the occurrence of biofilm deposition.
Clearly, biofilms can infiltrate our endoscopes; initial bedside cleaning, physical cleaning such as with the Pull Thru, and reprocessing are essential processes in removing biofilm and therefore the bacterial burden on and in an endoscope that will then transfer onto, or into, the patient.
Jane Townsend January 2019
References available [email protected]
|
|


INFORMED CONSENT
We know that all patients have the right to be fully informed regarding the procedures they are consenting to: including the risks, benefits, alternatives, and the risks associated with not going ahead.
Further than that, in order to make a truly informed decision patients need to understand what it means to them and their situation.
This right to an informed choice is part of the Health and Disability Commission’s Code of Rights.
It is agreed upon within the literature that informed consent is a process, not just the point in time when a patient signs the consent form.
It must begin well before the procedure, allowing the person to hear and digest information, seek clarification and answers to questions, and the time to consider it all and come to a decision.
We must remember that “showing up” having been referred does not denote informed consent.
Despite this acknowledgement healthcare research suggests that many people give consent to procedures and investigations that they do not actually understand; some without even reading the consent form.
Recently it was mooted that up to 60% of Americans undergoing healthcare procedures consented without reading the information contained within the consent form, with 44% having no idea what the procedure they were undergoing actually involved; with similar findings in other economically developed countries.
Additionally, many will simply agree to undergo a procedure as they believe it is not within their ability to make decisions about their health; preferring instead to be reliant on their healthcare provider.
"All patients have the right to be fully informed"
Decision making should be shared between the patient and the healthcare practitioner undertaking the informed consent process; it is imperative that the communication between the two is clear and appropriate, with both parties understanding their role.
The patient needs to ensure they understand what they are hearing, and if they don’t they must request further clarification.
The healthcare practitioner ensures that any miscommunication or diminished health literacy is acknowledged as soon as possible, and in such cases they must find better ways of conveying the information to the patient, including adjusting the content, format or language being used to suit the individual patient’s needs.
|
|
|
Many things can impact on people’s understanding of the information they are being given regarding healthcare procedures; English as a second language, fear and anxiety, maybe hearing or cognitive deficits can all contribute to decision making being confusing and difficult for patients.
Health literacy will also impinge on a person’s ability to become well informed, with many of the terms and words used being totally foreign, confusing, and often overwhelming. For some, simple terms are beyond their comprehension, with medical terminology and jargon seeming like another language.
Studies have indicated that information pamphlets accompanying the informed consent process need to be written in language gauged to be accessible by a 12-year-old in order to be readily accessible to all. This concept also applies to the content of the consent form itself; too often the forms are complicated and add to the confusion for the patient; simple terms and clear concise accessible language is necessary.
Providing information in a variety of ways can help, with suggestions explored in the literature including pamphlets with diagrams and lay terms in the person’s first language, and videos with animation and discussion regarding the procedure.
However, what research also states clearly is that none of these decision aids can replace the discussion with, and ability to ask questions of, the healthcare provider.
One suggestion to allow the patient the time to become informed and ponder their choices is to send out information packs within the weeks leading up to the procedure.
This pack could include the consent form itself. The literature agrees this is a good idea, so long as it is not expected that the consent form is then pre-signed without discussion with the healthcare practitioner; that is to say an information pack should never replace face to face communication within the informed consent process.
"Decision making should be shared between the patient and the healthcare practitioner"
One should never be expected to submit their consent for a medical procedure via an online system as again this leaves no ability for ensuring the consent is really an informed one as required ethically and by law.
One important aspect of modern endoscopy and informed consent is that in cases where polyps or other variables are found on surveillance the endoscopy can move from examination to treatment.
Gaining consent for the therapeutic treatment at this point is not possible.
Therefore, to avoid having to undergo two procedures, the consent process for gastroscopy and colonoscopy should involve a fully informed discussion around treatments that may take place based on findings throughout the examination.
Although it is ideally the practitioner undertaking the procedure that is involved in the consent process, in the case of endoscopy this responsibility can be delegated to another healthcare practitioner, such as a nurse, who is adequately versed in the procedure, the benefits, the risks, and the alternatives.
In these instances, patients must also be given the opportunity to speak directly to the practitioner undertaking the procedure should they wish.
In the field of endoscopy nurses are often involved in the informed consent process with the patient; with research suggesting that nurse gathered consent processes are more thorough and leave patients feeling more informed and empowered than physician gathered consents.
In conclusion:
The process of informed consent begins well before the procedure takes place and is a shared responsibility between the patient and their healthcare practitioner.
Whatever the procedure, whomever the patient, informed consent is a vital patient right within healthcare.
Without it, we risk suboptimal outcomes that impact on both the patient and the healthcare practitioner.
Jane Townsend January 2019
References available [email protected]
|
|

|
| |
|